04.23.13
Recently, the European Union introduced Directive 2010/32/EU for protection against injuries by sharp and pointed medical instruments through the use of needle protection systems, among other measures. Statistics show that needlestick injuries cause around one milltion work-related cases of health problems in Europe every year. The Needle-Trap syringe label by Schreiner MediPharm meets the requirements of the EU Directive and aims to contribute to a safe working environment in the hospital and healthcare sector.
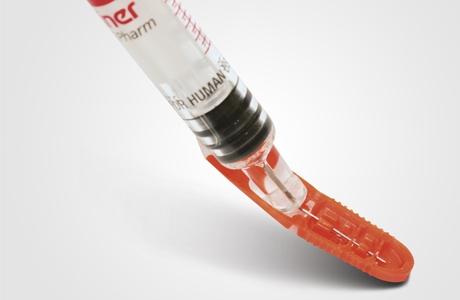 By May 11, member states have to implement Directive 2010/32/EU for protection against needlestick injuries into national legislation. Employers in hospitals and medical practice will then be obligated to analyze the risk of infection by blood or other body fluids when handling pointed and/or sharp instruments and to initiate appropriate actions. Employers will be required to provide processes or medical instruments with integrated protection mechanisms to reduce or eliminate the risk, in order to protect physicians, nurses, healthcare staff, and cleaning personnel as well as patients. The Needle-Trap needle protection system by Schreiner MediPharm was designed to help prevent needlestick injuries.
By May 11, member states have to implement Directive 2010/32/EU for protection against needlestick injuries into national legislation. Employers in hospitals and medical practice will then be obligated to analyze the risk of infection by blood or other body fluids when handling pointed and/or sharp instruments and to initiate appropriate actions. Employers will be required to provide processes or medical instruments with integrated protection mechanisms to reduce or eliminate the risk, in order to protect physicians, nurses, healthcare staff, and cleaning personnel as well as patients. The Needle-Trap needle protection system by Schreiner MediPharm was designed to help prevent needlestick injuries.
The syringe label with an integrated plastic trap secures the blood-contaminated needle after the injection. “The system is very easy to operate by healthcare personnel in a controlled and single-handed manner. After the needle has locked into the trap, it is irreversibly protected,” says Ann L. Merchant, president of Schreiner MediPharm. According to the company, there is no risk of infection due to potential blood splatters when activating the needle protection. An internationally recognized expert has demonstrated this as part of a scientific study. The quantities of blood (0.0001 μl) and medication content (0.00008 μl) released are hardly detectable and thus completely harmless.
Integration into the manufacturing process
Due to its compact design, Needle-Trap, like conventional labels, can be processed on standard labeling equipment with only minor modifications. Furthermore, the needle protection system is adaptable to all standard syringe dimensions and fits into common types of secondary packaging. Existing forms of packaging can be used without any modifications. This saves material as well as packaging and there is no additional space required for storage. Pharmaceutical manufacturers incur only minimal investment costs.
According to Schreiner MediPharm, the fast and easy integration into existing processes gives pharmaceutical manufacturers the possibility to quickly respond to the EU Directive. The innovative syringe label with integrated needle protection meets the stringent requirements of the U.S. Federal Drug Administration (FDA) for safety and quality and has been awarded FDA 510(k) clearance for marketing in the U.S.
Pharmaceutical corporations are already using Needle-Trap on millions of syringes, the company says. "Needle-Trap is an innovative and cost-efficient solution to significantly reduce the risk of needlestick injuries,” Merchant adds.
The syringe label with an integrated plastic trap secures the blood-contaminated needle after the injection. “The system is very easy to operate by healthcare personnel in a controlled and single-handed manner. After the needle has locked into the trap, it is irreversibly protected,” says Ann L. Merchant, president of Schreiner MediPharm. According to the company, there is no risk of infection due to potential blood splatters when activating the needle protection. An internationally recognized expert has demonstrated this as part of a scientific study. The quantities of blood (0.0001 μl) and medication content (0.00008 μl) released are hardly detectable and thus completely harmless.
Integration into the manufacturing process
Due to its compact design, Needle-Trap, like conventional labels, can be processed on standard labeling equipment with only minor modifications. Furthermore, the needle protection system is adaptable to all standard syringe dimensions and fits into common types of secondary packaging. Existing forms of packaging can be used without any modifications. This saves material as well as packaging and there is no additional space required for storage. Pharmaceutical manufacturers incur only minimal investment costs.
According to Schreiner MediPharm, the fast and easy integration into existing processes gives pharmaceutical manufacturers the possibility to quickly respond to the EU Directive. The innovative syringe label with integrated needle protection meets the stringent requirements of the U.S. Federal Drug Administration (FDA) for safety and quality and has been awarded FDA 510(k) clearance for marketing in the U.S.
Pharmaceutical corporations are already using Needle-Trap on millions of syringes, the company says. "Needle-Trap is an innovative and cost-efficient solution to significantly reduce the risk of needlestick injuries,” Merchant adds.












