04.26.18
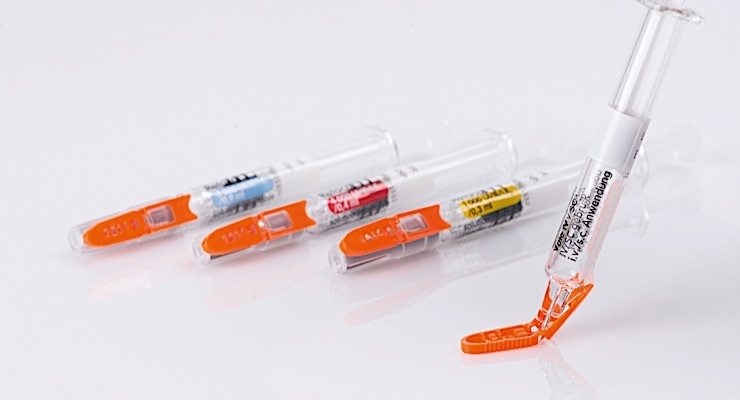
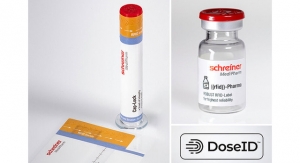
Syringes for Pfizer’s epoetin biosimilar have been outfitted with Needle-Trap needle protection labels from Schreiner MediPharm, a Germany-based global provider of specialty pharmaceutical labeling solutions for over 65 years. The pairing marks the first time Needle-Trap will be utilized with medication for the treatment of anemia. The newly equipped needles will begin usage in Germany and Austria, followed by a rollout on the broader European market.
Pfizer, one of the world's largest pharmaceutical companies, develops medicines and vaccines, as well as some of the most popular over-the-counter products. In search of a reliable solution to protect healthcare personnel from needlestick injuries when injecting the company’s epoetin product for the treatment of anemia, Pfizer chose Schreiner MediPharm's Needle-Trap. Pfizer has been using this label-based needle protection system on millions of syringes for a heparin drug since 2009.
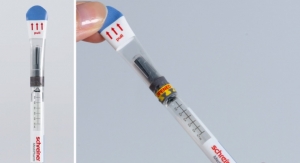
Needle-Trap features the following design: The plastic needle trap is an integral component of the label for prefilled syringes and serves to secure the needle after the injection has been performed. Due to its special design, it can be easily integrated into existing pharmaceutical manufacturing processes. Only minor modifications of the finishing and application equipment are needed, and there is no need for adaptation of secondary packaging. Minimal space during shipping, storage and disposal are required.
Schreiner MediPharm’s Needle-Trap meets the requirements of EU Directive 2010/32/EU, US NIOSH for safe instruments and DIN EN ISO 23908 for sharps injury protection. In addition, it has been cleared by an FDA 510(k) Premarket Notification in the United States.
A cost-effective alternative for prefilled syringes, Needle-Trap is intuitive due to its special design. It features simple and irreversible activation and requires no changes to the user’s injection technique as the needle protection is integrated into the syringe label.
Pfizer, one of the world's largest pharmaceutical companies, develops medicines and vaccines, as well as some of the most popular over-the-counter products. In search of a reliable solution to protect healthcare personnel from needlestick injuries when injecting the company’s epoetin product for the treatment of anemia, Pfizer chose Schreiner MediPharm's Needle-Trap. Pfizer has been using this label-based needle protection system on millions of syringes for a heparin drug since 2009.
Needle-Trap features the following design: The plastic needle trap is an integral component of the label for prefilled syringes and serves to secure the needle after the injection has been performed. Due to its special design, it can be easily integrated into existing pharmaceutical manufacturing processes. Only minor modifications of the finishing and application equipment are needed, and there is no need for adaptation of secondary packaging. Minimal space during shipping, storage and disposal are required.
Schreiner MediPharm’s Needle-Trap meets the requirements of EU Directive 2010/32/EU, US NIOSH for safe instruments and DIN EN ISO 23908 for sharps injury protection. In addition, it has been cleared by an FDA 510(k) Premarket Notification in the United States.
A cost-effective alternative for prefilled syringes, Needle-Trap is intuitive due to its special design. It features simple and irreversible activation and requires no changes to the user’s injection technique as the needle protection is integrated into the syringe label.