09.18.19
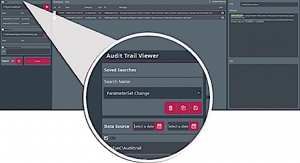
EyeC software takes into account the newest requirements for pharmaceutical labeling, dealing with data integrity, data security and audit trails. To complete this offer, the German company has launched the EyeC Audit Trail Viewer, new software that simplifies the preparation and review of audit trailing data. With the EyeC Audit Trail Viewer, responsible persons can now analyze and document process interventions for auditing in a convenient, controllable and comprehensive way. The software enables users to flexibly load and visualize trailing data and provides all the relevant information in a human-readable format as required by the US FDA Title 21 CFR Part 11. Various filter options enable refining entries to focus on critical events.
49-40-2263-5550, www.eyec.de
49-40-2263-5550, www.eyec.de