Greg Hrinya, Associate Editor09.17.18
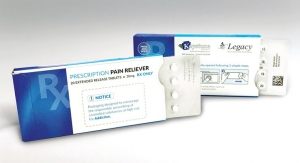
Perhaps more than any other market segment, quality plays a vital role in the success and functionality of pharmaceutical labels. In order to ensure quality and legibility, converters rely on the latest inspection equipment and software.
EyeC, with American facilities in Saint Charles, IL, offers a host of solutions to converters specializing in pharmaceutical labeling. These technologies must handle a wide range of products, too. Pharmaceutical labels often include braille, extended content labels, and plastic packaging.
“Quality requirements for pharmaceutical labels differ from those in other applications,” explains Oliver Peitzner, product manager at Eyec. “Any errors, like an illegible, incorrect or incomplete information on pharmaceutical labels, put patients’ lives in jeopardy and exposes pharmaceutical companies to a risk of litigation and a damaged reputation. Therefore, quality assurance is so essential for pharmaceutical packaging. Furthermore, contrary to other industries, pharmaceutical companies set the focus of their quality control mainly on text verification rather than on the mere appearance of printed labels.”
The integrity of a pharmaceutical label must also meet strict regulations and norms, such as ISO 9001, GMP, GAMP 5, and must guarantee validation according to the guidelines provided in US FDA Title 21 CFR Part 11 and comply with local directives.
Pharmaceutical companies require specific processes, such as standardized requalification procedures, for their inspection systems in order to guarantee consistent inspection results. Otherwise, they will implement specific workflows that allow them to fully document and record inspection processes, which ensures data integrity and security.
“Pharmaceutical companies have very strict requirements concerning their inspection systems,” says Peitzner. “Even minor defects, like the dot on top of an ‘I’ in a small font must be detected. Moreover, pharmaceutical companies want to have their inspection systems checked on a regular basis in order to ensure consistent inspection quality.”
EyeC validates the scanners in use with the software as a whole system. EyeC also has the ability to provide validation support with documentation, which would not be possible if only the software was delivered.
EyeC offers multiple products for pharmaceutical companies. With EyeC Proofiler Content, errors are detected early during the creation of the PDF file. Prepress studios can automate PDF profing processes with the help of the EyeC Proofiler Graphic and the EyeC Workflow Integration, allowing them to correct any problems even before producing plates. The scanner-based off-line inspection systems of the EyeC Proofiler series offer fast and secure testing of samples in various formats. Print shops use these systems for continuous sample testing during the printing process while pharmaceutical companies use them for a safe and precise incoming quality control.
The EyeC ProofRunner Label performs a 100% print inspection on the press or rewinder at maximum speed, notes Peitzner. For an even more effective print inspection process, the data from the press and rewinder can be linked with the EyeC Quality Link.
“The user-friendly EyeC inspection systems meet all data integrity requirements of the global pharmaceutical market and offer all possibilities for review of the AuditTrail,” says Peitzner. “Depending on the customer's requirements, special features such as integrating into workflows or connecting to database systems can ensure process security. For integration into pharmaceutical processes, EyeC offers a validation support package that facilitates validation.”
Peitzner also notes an important regulation change coming in February, which involves serialization. Printers and pharmaceutical companies must implement unique identifiers for each pharmaceutical product according to the IFA-Coding system. Companies then have to assign an explicit number to every single product package to enable an easy identification of each product.
“With the help of inline inspection systems, such as the EyeC ProofRunner, printers and pharmaceutical companies can check serialization and identify missing or duplicate data during printing and further processing,” adds Peitzner.
EyeC, with American facilities in Saint Charles, IL, offers a host of solutions to converters specializing in pharmaceutical labeling. These technologies must handle a wide range of products, too. Pharmaceutical labels often include braille, extended content labels, and plastic packaging.
“Quality requirements for pharmaceutical labels differ from those in other applications,” explains Oliver Peitzner, product manager at Eyec. “Any errors, like an illegible, incorrect or incomplete information on pharmaceutical labels, put patients’ lives in jeopardy and exposes pharmaceutical companies to a risk of litigation and a damaged reputation. Therefore, quality assurance is so essential for pharmaceutical packaging. Furthermore, contrary to other industries, pharmaceutical companies set the focus of their quality control mainly on text verification rather than on the mere appearance of printed labels.”
The integrity of a pharmaceutical label must also meet strict regulations and norms, such as ISO 9001, GMP, GAMP 5, and must guarantee validation according to the guidelines provided in US FDA Title 21 CFR Part 11 and comply with local directives.
Pharmaceutical companies require specific processes, such as standardized requalification procedures, for their inspection systems in order to guarantee consistent inspection results. Otherwise, they will implement specific workflows that allow them to fully document and record inspection processes, which ensures data integrity and security.
“Pharmaceutical companies have very strict requirements concerning their inspection systems,” says Peitzner. “Even minor defects, like the dot on top of an ‘I’ in a small font must be detected. Moreover, pharmaceutical companies want to have their inspection systems checked on a regular basis in order to ensure consistent inspection quality.”
EyeC validates the scanners in use with the software as a whole system. EyeC also has the ability to provide validation support with documentation, which would not be possible if only the software was delivered.
EyeC offers multiple products for pharmaceutical companies. With EyeC Proofiler Content, errors are detected early during the creation of the PDF file. Prepress studios can automate PDF profing processes with the help of the EyeC Proofiler Graphic and the EyeC Workflow Integration, allowing them to correct any problems even before producing plates. The scanner-based off-line inspection systems of the EyeC Proofiler series offer fast and secure testing of samples in various formats. Print shops use these systems for continuous sample testing during the printing process while pharmaceutical companies use them for a safe and precise incoming quality control.
The EyeC ProofRunner Label performs a 100% print inspection on the press or rewinder at maximum speed, notes Peitzner. For an even more effective print inspection process, the data from the press and rewinder can be linked with the EyeC Quality Link.
“The user-friendly EyeC inspection systems meet all data integrity requirements of the global pharmaceutical market and offer all possibilities for review of the AuditTrail,” says Peitzner. “Depending on the customer's requirements, special features such as integrating into workflows or connecting to database systems can ensure process security. For integration into pharmaceutical processes, EyeC offers a validation support package that facilitates validation.”
Peitzner also notes an important regulation change coming in February, which involves serialization. Printers and pharmaceutical companies must implement unique identifiers for each pharmaceutical product according to the IFA-Coding system. Companies then have to assign an explicit number to every single product package to enable an easy identification of each product.
“With the help of inline inspection systems, such as the EyeC ProofRunner, printers and pharmaceutical companies can check serialization and identify missing or duplicate data during printing and further processing,” adds Peitzner.