11.16.17
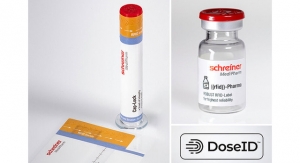
Schreiner MediPharm, the Germany-based provider of specialty pharmaceutical labeling products with a US facility in Blauvelt. NY, has introduced new features for its Autoinjector-Labels. The customizable solutions can now imcorporate an irreversible first-opening indicator with an anti-recapping functionality, as well as integrated dust protection for particularly challenging applications.
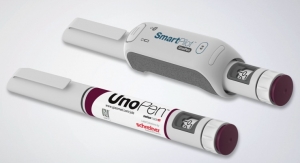
The market for injection devices such as autoinjectors and pens is rapidly growing. Factors in this expansion include the trend toward self-medication, the increasing incidence of chronic diseases, and a growing biotech sector. The sophisticated, innovative labeling solutions often needed in this sector drove Schreiner MediPharm to develop additional features for its Autoinjector-Labels.
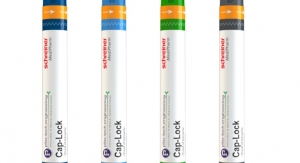
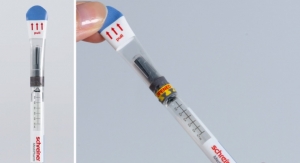
Schreiner MediPharm’s newest concept combines first-opening protection with an anti-recapping function and dust protection. The label wraps around the autoinjector, including the cap, providing effective dust and dirt protection before the device is initially opened. An integrated perforation clearly indicates that the label has been opened.
The label is positioned in a way that prevents undetected recapping of the autoinjector for illegal reuse. Other possible features include:
• A hologram for counterfeiting protection
• Inspection windows with an anti-UV varnish coating, to protect light-sensitive active drugs
• A room temperature indicator
• An anti-slip varnish for enhanced grip and safe handling
Injection treatment aids for such serious conditions as diabetes, rheumatoid arthritis, anaphylactic shock and multiple sclerosis need to be easy and safe in use, particularly for health- and motor-impaired patients. Schreiner MediPharm’s functional labels provide valuable assistance that address a variety of needs.
Schreiner MediPharm presented the novel label concept for the first time at the recent Parenteral Drug Association (PDA) conference Pre-filled Syringes and Injection Devices. Gene Dul, president of Schreiner MediPharm US, summarized the company’s commitment: “Our aim is to offer our customers and partners innovative and application-specific solutions which are optimally tailored to their needs, using the latest technologies to enhance product and patient safety. At the same time, we ensure that our sophisticated and functional label designs can be easily integrated into the pharmaceutical manufacturer’s production processes.”
The market for injection devices such as autoinjectors and pens is rapidly growing. Factors in this expansion include the trend toward self-medication, the increasing incidence of chronic diseases, and a growing biotech sector. The sophisticated, innovative labeling solutions often needed in this sector drove Schreiner MediPharm to develop additional features for its Autoinjector-Labels.
Schreiner MediPharm’s newest concept combines first-opening protection with an anti-recapping function and dust protection. The label wraps around the autoinjector, including the cap, providing effective dust and dirt protection before the device is initially opened. An integrated perforation clearly indicates that the label has been opened.
The label is positioned in a way that prevents undetected recapping of the autoinjector for illegal reuse. Other possible features include:
• A hologram for counterfeiting protection
• Inspection windows with an anti-UV varnish coating, to protect light-sensitive active drugs
• A room temperature indicator
• An anti-slip varnish for enhanced grip and safe handling
Injection treatment aids for such serious conditions as diabetes, rheumatoid arthritis, anaphylactic shock and multiple sclerosis need to be easy and safe in use, particularly for health- and motor-impaired patients. Schreiner MediPharm’s functional labels provide valuable assistance that address a variety of needs.
Schreiner MediPharm presented the novel label concept for the first time at the recent Parenteral Drug Association (PDA) conference Pre-filled Syringes and Injection Devices. Gene Dul, president of Schreiner MediPharm US, summarized the company’s commitment: “Our aim is to offer our customers and partners innovative and application-specific solutions which are optimally tailored to their needs, using the latest technologies to enhance product and patient safety. At the same time, we ensure that our sophisticated and functional label designs can be easily integrated into the pharmaceutical manufacturer’s production processes.”