Calvin Frost10.06.17
And a wonderful way to introduce this second column on green chemistry and biopolymers. What fascinates me – and I trust you – is the increasing interest and demand for these alternatives to synthetics. I believe I mentioned in my last column that the dollar volume of biobased products will grow to $10 billion by 2020 or before. That’s doubling this technology in five years or fewer. Below you’ll find out what I believe to be the key driver. In the meantime, what I didn’t tell you in the earlier column is the diversity of applications. Green coatings, for example, are growing by 5-7% a year, and this is expected to continue through 2020 because of the focus on stronger environmental regulations and a need to reduce VOC emissions. Sustainable inks, whether they be biodegradable, bio-renewable or eco-friendly, are replacing oil-based, synthetic polymers. Paint for a variety of applications, from homes to automobiles, is no longer oil based. All of these products are now latex (water) or modified acrylic/latex, another wonderful example of green chemistry working its magic. Both of these, inks and paints, are using friendly compounds derived from trees, plants, bugs, and so on. Ink, in particular, can include gums, resins, waxes, solvents, oils, and other polymers, but all are from natural sources. Biobased polyols are being used in powders and coatings also with natural ingredients, with the objective of achieving performance criteria while meeting sustainability goals. And, that’s the real key: balancing performance with sustainability. That word, balance, once again!
The use of biobased materials in coatings isn’t new. For example, shellac is based on a resin secreted from the lac bug (Mr. Editor, did you know that?). And, some of the first polyurethane chemistry was based on castor oil, which is: A vegetable oil obtained by pressing the seeds of the castor oil plant (Ricinus communis). The common name “castor oil,” from which the plant gets its name, probably comes from its use as a replacement for castoreum, a perfume base made from the dried perineal glands of the beaver (castor in Latin).
Years ago, when we started using biobased chemistry, performance was always the element that caused modification of our chemistry. This, of course, was before sustainability became a driver. In the early days, you could modify natural polymers and achieve your performance requirements by adding petrochemicals. However, when we add these polymers and create, for example, a polyester, there is a very strong possibility that we will create a product that is high in NOx or chlorine emission. That’s a real serious problem. In polymer chemistry (only for a moment), polyols are compounds with “multiple hydroxy functional groups available for organic reactions.” When you start changing carbon chains you create interesting polymeric structures. One of these, Azelaicacid, is commercially available and made solely from biobased feedstocks. Azebic acid is used as an additive in a variety of polyol applications such as a food sweetener. Suffice it to say, our polymer chemicals today are more focused on green chemistry than ever. The driver is the brand owner! The brand owner wants materials that do not create end of life problems. And all of this is driven by an increasing focus on external producer responsibility (EPR). Essentially, new and continuing growth in regulation and legislation is saying that if you make it you are responsible from cradle to grave. Biobased chemistry is essential to making EPR successful!
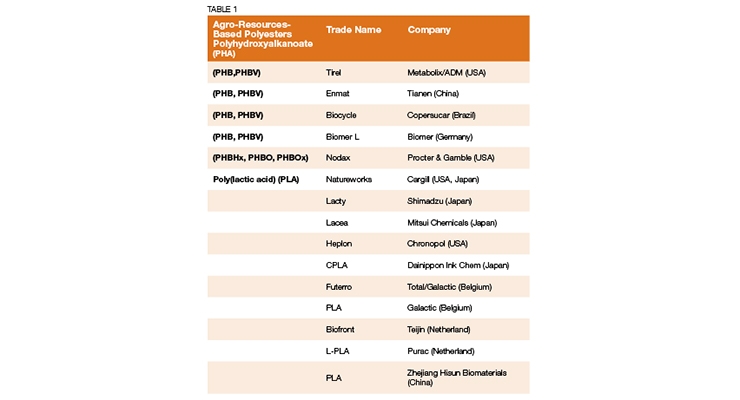
Please find below a list of acceptable polymers or materials for use from the website of Professor Luc Averons. These are all based from natural sources and I believe a good start for understanding the variety of green chemistries available today:
(See Table 1)
Professor Averons also has another chart that lists various physical properties from this same family of chemistries. These include density, melting point, tensile strength, biodegradability, etc. The point he is making is that today we can design biobased polymers, in this case, biobased polyesters, to give specific performance results.
Here are a few examples of some new bio-plastics applications:
Another Letter from the Earth
Calvin Frost is chairman of Channeled Resources Group, headquartered in Chicago, the parent company of Maratech International and GMC Coating. His email address is
cfrost@channeledresources.com.
The use of biobased materials in coatings isn’t new. For example, shellac is based on a resin secreted from the lac bug (Mr. Editor, did you know that?). And, some of the first polyurethane chemistry was based on castor oil, which is: A vegetable oil obtained by pressing the seeds of the castor oil plant (Ricinus communis). The common name “castor oil,” from which the plant gets its name, probably comes from its use as a replacement for castoreum, a perfume base made from the dried perineal glands of the beaver (castor in Latin).
Years ago, when we started using biobased chemistry, performance was always the element that caused modification of our chemistry. This, of course, was before sustainability became a driver. In the early days, you could modify natural polymers and achieve your performance requirements by adding petrochemicals. However, when we add these polymers and create, for example, a polyester, there is a very strong possibility that we will create a product that is high in NOx or chlorine emission. That’s a real serious problem. In polymer chemistry (only for a moment), polyols are compounds with “multiple hydroxy functional groups available for organic reactions.” When you start changing carbon chains you create interesting polymeric structures. One of these, Azelaicacid, is commercially available and made solely from biobased feedstocks. Azebic acid is used as an additive in a variety of polyol applications such as a food sweetener. Suffice it to say, our polymer chemicals today are more focused on green chemistry than ever. The driver is the brand owner! The brand owner wants materials that do not create end of life problems. And all of this is driven by an increasing focus on external producer responsibility (EPR). Essentially, new and continuing growth in regulation and legislation is saying that if you make it you are responsible from cradle to grave. Biobased chemistry is essential to making EPR successful!
Please find below a list of acceptable polymers or materials for use from the website of Professor Luc Averons. These are all based from natural sources and I believe a good start for understanding the variety of green chemistries available today:
(See Table 1)
Professor Averons also has another chart that lists various physical properties from this same family of chemistries. These include density, melting point, tensile strength, biodegradability, etc. The point he is making is that today we can design biobased polymers, in this case, biobased polyesters, to give specific performance results.
Here are a few examples of some new bio-plastics applications:
- Aimplos has developed a new sustainable package that extends the shelf life of cheese and fresh pasta, which meets the European Project Capitals BIO-4MAP. The result is a new barrier with a 25% lower cost than conventional membranes and has both a lower environmental impact and lower carbon footprint. “The layers of bioplastics (PLA, PVOH, and adhesives) and a wax coating from olive leaves, has made possible insulating the product from oxygen and humidity in order to avoid the growth of bacteria and fungi.”
- Sheffa Foods has developed a new 100% compostable wrapper for its “coffeehouse inspired granola bar.” The wrapper is a biobased and renewable flexible packaging substrate, designed to break down and fully return to nature, becoming part of the food waste stream.”
- Tufts University biomedical engineers are working on green chemistry that will answer the fast fruit rot dilemma. “Enquiries found that fruits can stay fresh for more than a week without refrigeration if coated in an odorless biocompatible silk solution. Fibroin, an insoluble protein found in silk, has the ability to stabilize and protect other materials while being fully biocompatible and biodegradable.” Their test fruit is strawberries. After being dipped in a solution of 1% silk fibroin, shelf life is extended by seven days, with no deleterious effect on the strawberries or the consumer.
Another Letter from the Earth
Calvin Frost is chairman of Channeled Resources Group, headquartered in Chicago, the parent company of Maratech International and GMC Coating. His email address is
cfrost@channeledresources.com.